Stay back! You have gone too close! This blog is purposefully created for chemistry students of Mr Kwok. Only discussion pertaining to chemistry is appreciated and welcomed. Now, start reacting. =)
Saturday, October 17, 2009
Consultation slots
Please take note of the consultation slots.
As there is a limitation in the upload of the file, the times on the second page is the same as the first page.
Consultation Schedule
For clarification, please SMS me. Take care!
Regards
Mr Kwok
Wednesday, October 14, 2009
Final Push - JC 2s
We are finally down to the last three weeks.
Week 3:(Starting 19th Oct): Wed and Thur.
Week 2:(Starting 26th Oct): Mon, Tues, Thurs and Fri.
Week 1:(Starting 2nd NovO: Tues, Wed, Thurs and Fri.
Week 0: ALL THE BEST. Contact me at my gmail.
Instructions:
Week 3: Wed - CH203 (Grp Kr & Xe) Thur - CH209 (Grp 4 & 5)
Week 2: Tues & Fri - for CH203 Mon & Thurs - for CH209
(In this week, Kr, Xe, Grp D and E will see me twice in this week. The remaining groups will see me once.)
Week 1: Tues & Fri - for CH203 Wed & Thurs - for CH209
(In this week, all groups will see me twice EXCEPT He, Ne, A and B.)
Group leaders of He, Ne, Ar, Kr, Xe, A, B, C, D and E. I have your names and I will be expecting you to sms me to tell me the days you are coming and whether you prefer AM or PM. I will SMS you the times and you will have to let me know whether it is agreed or not.
If you are not clear, do SMS or call if necessary. If you need more slots than the prescribed number, please also text me and allow me to know and see if I can make further arrangements.
Regards
Mr Kwok
Monday, October 12, 2009
Last week!
Please click here to download the following materials:
1. Click here for N04 Paper 3
2. Click here for N05 Paper 3
3. Click here for N08 Paper 2
Please print a copy for this week's class.
Regards
Mr Kwok
Wednesday, September 16, 2009
Homework before 23rd Sept 2009
 |
| From KWOK The Chem Teacher |
I am certain all of you are aware that pressure affects equilibria which has changes in the number of gaseous molecules. Hence, to ensure a constant pressure to be created is more difficult than to create an environment for constant temperature. Hence, please ponder on the above question. The solution isn't complicated but it is an application of what we have learnt.
Lastly, ensure your answers are clear and concise. This solution does not need excessive elaboration.
Background
The background of the question came from two different questions found in separate preliminary examination paper. The questions raised a curious thoughts, how is it possible to ensure a constant pressure for gaseous reaction system with temperature being kept constant?
Let's take Haber Process as the example. From its equation, you can tell that the number of particles at equilibrium will be less than the number of particles at the start of the reaction. Hence, if pressure was kept constant (i.e. inital P = final P), with a decrease number of particles, the volume must decrease. Therefore a contraction occurs.
Thus, the curious question. How is it possible for constant pressure, constant volume and constant pressure be maintained for the reaction for Haber Process? Fundamentally, the pressure exerted by the system has to be that exerted by the gas particles.
Hence, if initially we only have the reactants and the total pressure is 200 atm, how can a reduction in gas particles result in the pressure to still remain the same; when volume and temperature are constant? We would actually expect that pressure decreases.
Therefore, my suggestion is to add inert gas such as He into the reaction mixture. The inert gas added will ensure that the total pressure remains, in addition, this addition will not keep causing the equilibrium position to shift.
However, Jing Yew made an excellent suggestion of using a vortex. Personally, I am clueless to what it is. However, a check with a Physics teacher I learnt that it is a machine that can alter the kinetic energy of the gas particles. Hence, if this machine is feasible, it can alternate the speed of the particles at equilibrium to ensure that the total pressure remains the same as the initial pressure.
The other question
The simplest way is to cool the reaction mixture quickly and separate the species. Quickly because you do not want equilibrium to have any time to respond to the change in temperature. In addition, cooling ensure that the particles do not have enough energy to overcome Ea, hence hardly any forward or backward reaction can take place.
Subsequently, extract a small portion of the sample (to ensure only small amount of NH3 is present), then titrate against standardised concentration of HCl with a suitable indicator The small amount ensure that you will not need to have to use an exaggerated amount of HCl to attain full neutralisation.
Misconceptions
Some glaring misconceptions include
(1) Thinking that removing gases (especially NH3) will do the trick. That is a BIG NEVER. Since, the number of gas particles will decrease as we compare the initial with the equilibrium, removing particles will make the situation worse.
(2) Adding more gas reactants. Will that will ensure that you can get a constant temperature, but can you maintain the equilibrium? If you add a reactant, chances are equilibrium position will shift right. Hence, you will constantly have a situation where equilibrium position shifts and hence you can't achieve the equilibrium.
(3) Using a pH to determine the concentration of ammonia present. That is extremely inaccurate! We never do that!
(4) Apply PV = nRT. I am now sure how are u going to separate the ammonia from the mixture and hence measure the partial pressure of ammonia. Subsequently, using the ideal gas equation to calculate number of moles of ammonia.
Conclusion
Personally, I am not sure if this is what happen in industries. However, from my extensive search on the web, my sense is probably Jing Yew may be more accurate than I am. Since, the gases in Haber process are actually injected into the reaction vessel at a particular speed to create the desired pressure (Note: More KE the gas particles has, the greater force they exert when the collide of the vessel's wall). The notion of speed causing the pressure gives me that suspicion that it could be a voltex or at least some instructment that can adjust the speed of the particles and yet ensuring the volume, pressure and temperate are kept constant.
Thursday, September 10, 2009
Term 4 Plans
I hope that you had a good break. Please work on the following as preparation for the new term.
(1) Work on the revision package given you.
(2)
(3) Go through your prelim questions again.
When term re-starts, we will go through,
(1) Prelim questions.
(2) TYS papers. Expect P3 to be covered and remaining papers would be on your own.(Probably, 04 till 08, depending on time.)
(3) Selected 2009 prelim papers.
Regards
Mr Kwok
P.S. This is updated. Please spread the news around.
Saturday, September 5, 2009
Thanks!
Thank you for your good wishes, gifts and kind words. :)
Sent via BlackBerry from SingTel!
Wednesday, July 15, 2009
Periodicity - Electrical Conducitiy Trend
The diagram below depicts the electricial conductivity across Period 3.

In order for us to explain the trend, we need to know the structure the substance exists as. If the substance is a metal, it will have a sea of delocalised electrons available which acts as mobile charge carriers for electricity to be conducted. Thus, simplistically more available the sea of electrons, the better the conductivity. Therefore, structure provides important information to whether there are any charge particles which can move easily.

Interestingly, the explanation to why Si acts as a semi-conductor can be found here. But it is not part of the "A" level syllabus. If you can appreciate the article, you may wish to think about what is the more accurate representation of metallic bonds.
Lastly, there is a slight anonamlly. In this post, I accounted that Al has a higher electrical conductivity as it has a greater pool of delocalised electrons as Al has more valence electrons - Ironically, this contradicts the reasoning to why Al's melting point is not much higher than Mg. However, the details of this reasoning is beyond the scope of this discussion.
-- -- -- -- --
Article written by Kwok YL 2009.
- If you would like to use this source, kindly drop me a note by leaving behind a comment with your name and institution. I am all for sharing as the materials on this blog is actually meant for the education purpose of my students.
- This material is entirely written by the author and my sincere thanks will be given to anyone who is kind, generous and gracious to point out any errors.
Periodicity - Ionisation Energy Trend
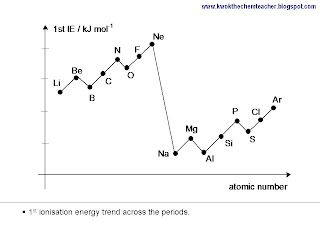
Generally, as we move across the period, the 1st ionisation energy increases. This is because despite an electron is added, the number of inner shells remains the same hence the shielding effect is relatively constant. While the nuclear charge increases. Therefore, the combination of these two reasons result in an overall increase in the 1st ionisation energy.
There are a couple of dips seen and these are exceptions to the trend. Generally, the dips are present because the causes for the dips are more significant than the effect due to increase in effective nuclear charge. The mind map below provides you with a structure which will explain the trend, including the exceptions.

In addition, there is another explanation to why the ionisation energy differs between period. We move from Period 2 to Period 3, the size of the atoms get larger. The added inner shell of electrons results in the valence shell of elements in Period 3 to be further away from the nucleus. Hence, electrostatic attraction between nucleus and valence shell becomes weaker, therefore 1st ionisation energy is smaller.
Article written by Kwok YL 2009.
- If you would like to use this source, kindly drop me a note by leaving behind a comment with your name and institution. I am all for sharing as the materials on this blog is actually meant for the education purpose of my students.
- This material is entirely written by the author and my sincere thanks will be given to anyone who is kind, generous and gracious to point out any errors.
Monday, July 13, 2009
Periodicity - Melting Point Trend.
Across the period from Na to Ar: Na, Mg and Al exist as giant metallic structures. While Si exists as giant covalent molecule (macromolecular structure). While P, S, Cl and Ar exists as simple discrete molecules. These elements' standard states formula of P4, S8, Cl2 and Ar respectively.
Hence, to explain the melting point trend across the period, it is recommended to define the structures the substance exists as first before proceeding further. The following diagram constructs the explanation needed.

Lastly, Al contains 3 valence electrons. Although these 3 should contribute to the sea of electrons, it does not happen because it will be very difficult for Al to lose so many electrons to contribute to the sea of electrons. Therefore, the increase in melting point from Al to Mg is much smaller than the increase form Mg to Na.
-- -- -- -- --
Article written by Kwok YL 2009.
- If you would like to use this source, kindly drop me a note by leaving behind a comment with your name and institution. I am all for sharing as the materials on this blog is actually meant for the education purpose of my students.
- This material is entirely written by the author and my sincere thanks will be given to anyone who is kind, generous and gracious to point out any errors.
Sunday, July 5, 2009
Electrochemistry - Calculation of Ecell.
(1) Predict which is the species that will be oxidised.
(2) Predict which is the species that will be reduced.
(3) Write the half-equation for the species that is oxidised; ensuring that it is found on the RHS.
(4) Write the half-equation for the species that is reduced; ensuring that it is found on the LHS.
(5) Apply anti-clockwise rule to calculate Ecello
The video below takes you through the above steps, demonstrate how the Ecello is calculated for a reaction between Zn and Cu2+ .
Lastly, when calculating the Ecello, please be mindful that we assume that the standard condition of 1 moldm-3 was used. However, when a smaller (or different) concentration is used, the reduction potential of the species is affected because of Le Chatelier's Principle as the equilibrium is affected.
-- -- -- -- --
Article written by Kwok YL 2009.
- If you would like to use this source, kindly drop me a note by leaving behind a comment with your name and institution. I am all for sharing as the materials on this blog is actually meant for the education purpose of my students.
- This material is entirely written by the author and my sincere thanks will be given to anyone who is kind, generous and gracious to point out any errors.
Friday, July 3, 2009
Chemical Energetics - Applying Gibbs Free Energy

(1) Predict the temperature for reactions to happen or become spontaneous.

(2) Predict the temperature for reactions to become non-spontaneous.

(3) Units.
-- -- -- -- --
Article written by Kwok YL 2009.
- If you would like to use this source, kindly drop me a note by leaving behind a comment with your name and institution. I am all for sharing as the materials on this blog is actually meant for the education purpose of my students.
- This material is entirely written by the author and my sincere thanks will be given to anyone who is kind, generous and gracious to point out any errors.
Thursday, July 2, 2009
Homework - Change in venue.
There is a new online assignment, please read the following:
Usage of Google Site - Ground Rules
Regards
Mr Kwok
Monday, June 22, 2009
Chemical Energetics - Gibbs Free Energy
With the introduction of entropy; which indicates the distribution of particles in a system does not help us to predict whether a reaction occurs or not. This is because we do observe reactions which show an increase in disorder (positive entropy change) and reactions which show a decrease in disorder (negative entropy change) happen.
However, we do observe that certain reactions, which has a certain enthalpy change and certain entropy change, can take place at certain temperature but will be unable to do so at other temperatures.
This observation allows for the introduction of Gibbs free energy. The change in Gibbs free energy accounts for the change in enthalpy and the change in entropy. It is this term, when negative in value, tells us that the reaction will be spontaneous, while when it is positive, tells us that the reaction is non-spontaneous. This term is a direct application of 2nd Law of Thermodynamics.
 |
In addition, it is vital that we are able to appreciate that by changing temperature, it can sometimes cause a chemical reaction which was initially spontaneous to become non-spontaneous as we can see from the diagram below.

It is good to take note that when temperature change, when we apply the above formula we assume that the enthalpy change and entropy change remains the same despite there is a change in temperature. (Or at least, that the change in entropy and change in enthalpy due to temperature cancels each other out.)
However, the change in temperature may lead to the substances (in the chemical equation) to be in a different phase at the new temperature as compared to the old one (An example is shown below). This phase change results in a significant change in the entropy and hence we cannot assume that the change in entropy remains constant at a different temperature.
While at the "A" level Chemistry syllabus, we do assume that enthalpy change remain the same despite temperature change. This is because the assumption is that the enthalpy change of the reaction is affected solely by the chemical bonds present in the substances and that these bonds remain of the same strength even at a higher temperature. - (Although, I must add that this assumption is used to simplify the calculation of enthalpy change.)
In conclusion, reaction that is spontaneous will happen on its own in nature, while reactions that are not spontaneous will need an external supply of energy so that it can take place - for example photosynthesis is one such process, thus planets make use of light energy to enable this reaction to take place.
-- -- -- -- --
Article written by Kwok YL 2009.
- If you would like to use this source, kindly drop me a note by leaving behind a comment with your name and institution. I am all for sharing as the materials on this blog is actually meant for the education purpose of my students.
- This material is entirely written by the author and my sincere thanks will be given to anyone who is kind, generous and gracious to point out any errors.
Friday, June 5, 2009
Chemical Energetics - Entropy
Introduction
 Hence, if there are more ways the particles can be arranged (as seen above), the more disordered and naturally, the system is more randomed. Since, the units of entropy is J mol-1 K-1, we would expect a energetics way to describe entropy (Just like potential energy describes the electrostatic attractive forces and etc).
Hence, if there are more ways the particles can be arranged (as seen above), the more disordered and naturally, the system is more randomed. Since, the units of entropy is J mol-1 K-1, we would expect a energetics way to describe entropy (Just like potential energy describes the electrostatic attractive forces and etc).Entropy, thus, can be seen as how spread out the energy possessed by the system (is it localised or not). To fully appreciate this idea, it is important for someone to know the concept of molecular orbital theory, which is beyond the scoop of this discussion.
Therefore, the main tenet of this post will be using the description that entropy can be determine by the number of ways the the particles can be arranged. Thus, using this as a means to account for how the following facts can cause entropy to increase.
(a) Change from Solid to liquid (change from liquid to gas).
The idea of solid is that the volume is fix and there is a definite structure. Hence, the arrangement of the particles is fixed thus very little different distribution of the arrangement of particles since they aren't mobile.
In liquids, the volume is definite, however the structure isn't. Hence, this helps to conceptualise that the arrangement of particles in the liquid state is less rigid; there is a more varied arrangements of the particles because there is some amount of mobility.
Lastly, the volume of a gas is not definite (since gas can be compressed), and there is no logical means to describe its shape, hence, there must be numerous arrangementa of the particles in gasous state, since they will have the greatest mobility.
Hence, entropy increases when we change the physical state of a substance from solid to liquid (and from liquid to gas).
 (b) Increasing in Temperature
(b) Increasing in TemperatureGenerally increasing temperature will definitely cause the particles to gain mobility, since the kinetic energy which the particle possess increase and it would be more abled to overcome attractive forces that restrict its movement.
Hence, increasing temperature will certain aid to create a more varied distribution of the particles, and that causes entropy of the system to increase. However, the quantitative extent which entropy increase is beyond the scoop of this discussion.
 (c) Mixing
(c) MixingThe idea of mixing is when a solute is added to a solvent. In particle terms, in order to dissolve the solute in a solvent, the solute particles must be surrounded by the solvent particles.
For example, to dissolve glucose (solute) into water (solvent), the water molecules must surround the glucose molecules. Thus, to have water molecules to surround the glucose molecules favourably, water molecules must be able to form hydrogen bonding with the glucose molecules.
Needless to say, when we initially only have pure solute and pure solvent, the particles are concentrated amongst themselves. When mixing is done, it would result in a greater variety in the arrangement of the particles since they ceased to be concentrated amongst themselves (because of the concept of mixing). Hence, entropy increases when we mix substances together.
 (d) Production of (Gasous) more particles
(d) Production of (Gasous) more particlesThe production many small molecules can generally to cause an increase in entropy, which sometimes is the reason for causing endothermic reactions to be spontaneous.
However, it because of an increase in the total number of particles (after a chemical reaction) which results in entropy increase. This is because this creates a larger number of possible arrangements of the particles, hence an entropy increase.
In addition, an increase in number of gasous particles after a reaction, will most definitely cause entropy to increase, since gas particles are highly disordered, it will add to randomness of the system.
Therefore, it is also good to check the physical state which the products formed exist in, since formation of more solid products will not be expect to lead to an increase in entropy.

- If you would like to use this source, kindly drop me a note by leaving behind a comment with your name and institution. I am all for sharing as the materials on this blog is actually meant for the education purpose of my students.
- This material is entirely written by the author and my sincere thanks will be given to anyone who is kind, generous and gracious to point out any errors.
- Due to limitation of the blog and my limited code knowledge, I am unable to do superscripts and subscripts, hopefully in time to come, I would be able to do so.
Wednesday, June 3, 2009
Announcement
I hope that your June break is starting to be productive. "A" Levels is approaching (HOOOOWWW????).
This week's online review would be:
(1) Read through all the bonding posts. They can be found here.
(2) Watch the different videos to construct energy cycles, Born Haber Cycle (and energy level diagram) and oxidation number method. Go to the links, it will bring you to the summary page for the videos that I have.
What I will be expecting you to finish when I see you in July.
(1) Set A, B and C. - This includes all the topic. It is a compilation of Prelim papers.
(2) Physical Chemistry Package - This contains just physical chemistry questions.
For (2), please attempt on bonding, energetics and kinetics questions. If you have time, it would be excellent if you attempt electrochemistry and equilibrium. I will be posting materials on this blog, but it will not be so soon.
If you have any question, please drop me a message in the chatbox.
Regards
Mr Kwok
Tuesday, June 2, 2009
Chemical Energetics - Definitions
(A) First set:
 (1) In simple terms, the enthalpy change of reaction is per mole of reaction. What this implies is that the thermochemical equation is balanced and all the stoichiometric coefficients are intergers.
(1) In simple terms, the enthalpy change of reaction is per mole of reaction. What this implies is that the thermochemical equation is balanced and all the stoichiometric coefficients are intergers.(2) In the enthalpy change of formation, do note that the non-metal substances which exist as simple discrete molecules such as hydrogen and oxygen, exist as H2 and O2 respectively.
(3) As combustion are exothermic reactions, we will expect all enthalpy change of combustion to be exothermic.
(B) Second set:
 (4) The enthalpy change of atomisation stresses on the formation of one mole of gaseous atoms. Please do not be confused with this definition and that of enthalpy change of formation.
(4) The enthalpy change of atomisation stresses on the formation of one mole of gaseous atoms. Please do not be confused with this definition and that of enthalpy change of formation.(5) The second ionisation energy simply refers to the second electrons removed.
(6) While the second electron affinity refers to the second electron added.
(7) Lattice energy describes the strength of an ionic bond. It is directly proportional to the product of the charges of the cation and the anion and it is inversely proportional to the sum of the respective radius of the cation and anion. As lattice energy talk about formation of the ionic bond only, it is an exothermic process.
(C) Third set:
 (8) Enthalpy change of solution is the enthalpy change when one mole of substance dissolves in water. We cannot assume that dissolving substance into water does not produce or require heat.
(8) Enthalpy change of solution is the enthalpy change when one mole of substance dissolves in water. We cannot assume that dissolving substance into water does not produce or require heat.(9) Enthalpy change of neutralisation stresses on the formation of one mole of water in a reaction between an acid and a base. Between a strong acid and a strong base, the enthalpy change of neutralisation is -57 kJ mol-1.
However, if a weak acid and a strong base is used (or vice versa), the enthalpy change of neutralisation is less exothermic than -57 kJ mol-1. This is because some amount of heat is required to dissociate the weak acid (or weak base) completely.
(10) Bond energy - do take note that it is defined to be the breaking of one mole of a given covalent bond to give gaseous products.
Hence, in order to ensure it is the energy that breaks the covalent bond, the substance must exist as a gas first. Generally, substances that have covalent bonds exist as simple discrete molecule. Hence, if liquid state or solid state is used, added energy is required to break the intermolecular forces. Therefore, the gas state is used as the intermolecular forces between gas molecules are the weakest.
(D) Final set: This provides additional information.

-- -- -- -- --
Article written by Kwok YL 2009.
- If you would like to use this source, kindly drop me a note by leaving behind a comment with your name and institution. I am all for sharing as the materials on this blog is actually meant for the education purpose of my students.
- This material is entirely written by the author and my sincere thanks will be given to anyone who is kind, generous and gracious to point out any errors.
Friday, May 29, 2009
Homework 8 (due Tuesday 2nd June 2009)
This application question may be slightly more challenging but it has to do with chemical bonding.
If you noticed SiO2 does not exists like CO2, where we obtain an SiO2 with a pair of Si=O bond.
Interestingly, Phosphorous prefer to exists as P4, with single bonds linking the phosphorous atoms while Nitrogen exists as N2 where there is a triple bond between the two N atoms.
You may wish to read about this (an article which you will learn in JC 2). If you understand that article, you will be able to explain for the above situation.
Regards
Mr Kwok
P.S. This is an interesting question to develop thinking skills. The chemistry concept of this will be largely covered in 2010.
Suggested Answer:
In CO2, the C=O bond is made up of a sigma bond and a pi bond. If you have read the hybridisation article, you would realise that for carbon has to undergo hybridisation of the 2s and 2p orbitals to form suitable hybrid orbital for the double bond. Since CO2 contains 2 C=O, the carbon is sp hybridised. Hence, the sp hybridised orbitals overlap with the 2p orbital of oxygen to get the sigma bond, while the 2p orbital carbon overlap in a side-on manner with 2p orbital of oxygen to get the pi bond.
In SiO2, it is just sigma bonds. Hence, silicon has hybridised its 3s and 3p orbitals to give sp3 orbitals, which are used for head-on overlapping with the p orbital of oxygen.
Hence, it is quite clear that the formation of pi bond is what is different in both cases. As the formation of pi bond between C and O is favourable, hence carbon uses the sp hybridised orbitals. The reason why pi bond between C and O is favourable is because the p orbital of C and that of O are able to effectively overlap with each other.
Usually large period 3 and below elements find it harder to form pi bonding. Their p orbital usually prefer not to have side-on overlap with the 2p orbital of small atoms such as O as the overlapping may not be sufficiently effective for the particular type of hybridisation to take place.
It is this reluctance to form pi bond between atoms that results in phosphorous to exist as P4 instead of P2. The former allows each P atom to form 3 bonds (hence satisfying their octet requirements), while the latter forces each p orbtial to contribute two 3p orbitals for side-on overlapping for pi bond. This side-on overlapping is not sufficiently effective and hence the hybridisation to get sp or sp2 is not favoured. Instead a sp3 is used to obtain sigma bonds between P atoms.
Comments:
I hope that you found this entry abit interesting. It is probably a "S" paper Chemistry question, but it is really testing us whether we understand why bonding take place between two atoms and why different types of bond can occur between the two atoms.
I shall also publise all your contributions and perhaps you could take time and compare the difference in how the thought processes were formulated.
Commendable Student's answers:
Please read Johanan's and Brandon's input. Jing Yew's contribution is interesting but it did not hit what I feel is the key point. The same should be said about Lyria's answer, which was coherent but I was not convinced that she understood the key point about how easy was it for the pi bond to be formed.
Wednesday, May 27, 2009
Chemical Energetics - More Energy Cycles
Like in the earlier post, the state symbols of all the substances have to be shown.
My example which I will use to illustrate how to draw the Born Haber cycle is the standard enthalpy change of formation of CaF2. As standard conditions are employed, the enthalpy change of atomisation of Ca and F are also in standard conditions. (You would expect enthalpy change of atomisation of solid F2 to be more endothermic than that of gaseous F2.)
It is interesting to note that it is quite impossible to obtain a standard condition for ionisation energy (I.E.) and electron affinity (E.A); we shall however assume that these will be constant at all temperatures.
(i) Born Haber Cycle:

(ii) Energy Level Diagram:

-- -- -- -- --
Article written by Kwok YL 2009.
- If you would like to use this source, kindly drop me a note by leaving behind a comment with your name and institution. I am all for sharing as the materials on this blog is actually meant for the education purpose of my students.
- This material is entirely written by the author and my sincere thanks will be given to anyone who is kind, generous and gracious to point out any errors.
Saturday, May 23, 2009
Homework 7 (due 25th May 2009)
Please copy/print out the following questions. Answer them on writing paper and submit your scripts on Monday.


Regards
Mr Kwok
Wednesday, May 13, 2009
Annoucement
Please read ALL the post on Chemical bonding and they can be found here.
Please read through ALL the answers to the homework question on bonding and they can be found here.
In addition, I would like to clarify how to draw the hydrogen bonding which cause ethanoic acid to dimerise in benzene. I think my answers in tutorial was alittle sloppy hence please use this one.
 Lastly, please encourage your classmates to read all these as part of the preparations - Let's keep our content updated and sharpen our skills to answer questions! And yes, this is your assignment this week.
Lastly, please encourage your classmates to read all these as part of the preparations - Let's keep our content updated and sharpen our skills to answer questions! And yes, this is your assignment this week.
Let's cheer for Ben and Xing Ting this Friday!
*Update 16th May: There is a new picture added and some re-organisation of the bonding posts.*
Regards
Mr Kwok
Sunday, May 10, 2009
Chemical Bonding - Application of IMF (part 2)
The basic tenet of solubility of molecular substance is "likes dissolve likes". In this phrase, it highlights that in order for a molecular substance to dissolve in a particular solvent, the intermolecular forces formed between the solvent molecule and the solute molecule must be stronger than/ or identical the intermolecular forces between solvent molecules only and between solute molecules only.
Thus, it is expected that polar molecular substance dissolves in polar solvents as similar type of intermolecular forces is formed. And for the same reason, non-polar molecular substances dissolve in non-polar solvent.
However, in the case of solubility of substance in water, we need to be abit more careful. A substance can be soluble in water if it can form hydrogen bonding with water. It need not be able to have hydrogen bonding on its own, but it definitely must be able to form hydrogen bonding with water. This is illustrated by the example below.

The formation of ion-dipole interaction is also the reason the reason to why ionic compounds prefer to dissolve in water than in a solvent which only has pd-pd (e.g. CHCl3). This is because solvents that can form hydrogen bonding forms the strongest ion-dipole interaction as compared to other solvents which can only exhibit pd-pd.
Lastly, a mind map is available to help you summarise this prose. This map will be a useful guide to conceptualising how the intermolecular forces is applied to predict solubility of a molecular solute.
-- -- -- -- --
Article written by Kwok YL 2009.
- If you would like to use this source, kindly drop me a note by leaving behind a comment with your name and institution. I am all for sharing as the materials on this blog is actually meant for the education purpose of my students.
- This material is entirely written by the author and my sincere thanks will be given to anyone who is kind, generous and gracious to point out any errors.
Saturday, May 9, 2009
Chemical Bonding - Application of IMF (part I)
In an earlier post, I briefly described to you the different intermolecular forces. In this write up, I will aim to show how the knowledge of intermolecular forces can be applied to account for the different boiling point trend (or melting) in a series of simple discrete molecules.
Do note that melting point can also be affected by how the molecules are packed. Smaller molecules can be packed more densely and hence when melting them, more intermolecular forces are broken and thus giving a larger melting point despite its intermolecular forces is weaker.
(A) Comparing id-id against pd-pd against Hydrogen bonding
We will compare the different types of intermolecular forces when (1) The Mr of the molecules is relatively similar and (2) The shape of the molecule is relatively the same.
In (1) this implies that the number of electrons in the series of compounds is the similar and hence id-id (which is dependent on number of electrons) will be similar. In (2) it will ensure that the electron cloud size is similar (hence id-id) is similar.
When this happens, Hydrogen bonding is the strongest while id-id is the weakest.
(B) Comparing the Mr of the molecule
When the a pair of molecules respective Mr is very different. This implies that the number of electrons the present in the pair is also very different. Hence, the size of the electron cloud will be different too.
Hence, this result in the larger molecule to be polarised easily and hence its induce dipole moment is larger and hence its id-id interaction is stronger and therefore having a higher boiling point.
(C) Comparing the shape of the molecule (e.g. Branched or unbranched)
Unbranched molecules such as CH3CH2CH2CH3 have an elongated electron cloud which is easier to be polarised. Hence, its induced dipole moment will be large and hence, id-id interaction is stronger. While unbranched molecules such as (CH3)4C have a spherical electron cloud which is less easily polarised and hence smaller induced dipole and thus weaker id-id.
(D) If Mr, shape of molecules and IMF are similar/identical
For both molecules satisfy this subtitle and both have hydrogen bonding. Then there are two main factors. (1) Extensive-ness of hydrogen bonding. For example H2O has more extensive hydrogen bonding than HF and NH3 because it has the best (most equal) lone pair to hydrogen ratio. (2) Using the definition of Hydrogen bonding as illustrated by this diagram, HF has a stronger hydrogen bonding than NH3 because F is more electronegative than N and hence making the d+ on H in HF to be larger.
When both molecules satisfy this subtitle and both have pd-pd interactions. There are also two main factors. (1) Electronegativity of the atom. More electronegative atom results in a larger dipole moment. (2) More electronegative atoms which create a dipole moment in the same direction. This will result in the molecule to have a larger overall net dipole moment.
Needless to say, when both have only id-id interaction, the boiling point should be the same.
As this is a length entry. A mind map, which shows you how to sequence your thoughts to construct the concise and comprehensive argument is found here.
-- -- -- -- --
Article written by Kwok YL 2009.
- If you would like to use this source, kindly drop me a note by leaving behind a comment with your name and institution. I am all for sharing as the materials on this blog is actually meant for the education purpose of my students.
- This material is entirely written by the author and my sincere thanks will be given to anyone who is kind, generous and gracious to point out any errors.
Friday, May 8, 2009
Chemical Bonding - Intermolecular Forces (mindmap)
You may want to click here to see the entire map.
-- -- -- -- --
Article written by Kwok YL 2009.
- If you would like to use this source, kindly drop me a note by leaving behind a comment with your name and institution. I am all for sharing as the materials on this blog is actually meant for the education purpose of my students.
- This material is entirely written by the author and my sincere thanks will be given to anyone who is kind, generous and gracious to point out any errors.
Thursday, May 7, 2009
Homework 6 (due before 11th May 2009) (part 2)
This is the second question.
Suggest an explanation to why XeF2 exists but NeF2 does not. [Hint: You are required to use the concept of atomic structure and chemical bonding in your answer]
Answer to Question 2
Xe is a Period 5 element and although it has 8 valence electrons, it has available empty d-orbital which can be used to expand the octet for additional bonding. While Ne is a Period 2 element and does not have available d-orbtial which can be used to expand the octet.
In additional, to expand the octet in Xe, it requires the unpairing of the valence electrons and exciting them to the d orbital, since in a same shell the d orbital has a higher energy than the s orbital and the p orbital. Thus, this rearrangement of electronic configuration requires energy. Noticeably, this energy required is compensated by the Xe-F bond that is formed (bond formation releases energy). Therefore, XeF2 exist.
Even if Ne can expand its octet, Ne's valence electrons are so close to the nucleus and thus experience strong electrostatic attraction. Thus to unpair them and move it to a higher energy orbital, you will need so much energy that even the formation of the Ne-F bond is unable to compensate the energy required use to rearrange the electrons configuration.
Comments for Question 2
1. If you have submitted the work, a mark is awarded to you. If you have mentioned expansion of octet due to available d orbitals or available orbitals a second mark is given to you. However, if you mentioned 3d orbital, you will not get the mark.
2. All of you had problems in getting the 3rd and 4th mark. I guess one has to realise that in order to expand the octet for bonding, there is a need to rearrange the electronic configuration of Xe, to make the electrons unpaired. This gives the 3rd mark. One of the unpaired electrons will occupy a higher energy orbital such as the d orbital and the unpair electron awaits to be bonded with F. (Note that formation of Xe-F bond makes use of equal sharing of electrons, where Xe and F each contribute one electron. Hence, you will need Xe to unpair its electrons pair.)
3. Finally, because the Xe-F bond is formed (bond formation is exothermic or energy releasing - this was mentioned in an earlier blog post), the energy release can compensate the energy required to rearrange the electronic configuration. This is the 4th mark.
4. Hence, to expand the octet these are the available reasons. Hence, sometimes when we want to consider about expansion of octet, the type of bond formed will determine if the expansion is favourable. Hence, if a strong covalent bond can be formed, we will get expansion of the octet. For this reason, XeBr2 does not exist as the Xe-Br bond is not sufficiently strong.
Homework 6 (due before 11th May 2009) (part 1)
There are two questions in today's assignment. Please copy question 1 and answer it on a piece of writing paper. Submit you answers before Tuesday's assembly.
Question 1 - Done in writing paper.
(a) Draw the dot and cross diagrams of (i) N2O (ii) O3 (iii) HNO3 (iv) H3PO4.
[Hint: (iii) and (iv) are mineral acids. As they are mineral acids they have at least one O-H.]
(b) Draw the lewis structure of H2O2. On this diagram, label all bond angles. In addition, determine the shape of H2O2.
(c) XeF2 is one of the most stable Xenon compounds. (i) Draw the dot and cross diagram of XeF2. (ii) Predict with explanation which bond is stronger Xe-F or He-F.
(d) Suggest an explanation for the following suitations. (i) MgCl2 has stronger interatomic bonds than NaCl. (ii) Given that the bond energy of C-Cl is 328 kJmol-1 and while the C-N is 292 kJ mol-1.
[Marks allocation for Q1: 3 + 4 + 4 + 4]
Suggested Solutions


 Comments
Comments(1) Becareful with your explanation in (c)(ii) - It cannot be a contradiction.
(2) Becareful with your explanation in (d)(ii) - It cannot contradict the given data. Hence, atom size for effective overlap isn't suitable. Lone pair repulsion is irrelevant as C does have that.
Chemical Energetics - Construction of cycle
In this entry, there are two videos available. The first video provides a simplified guide to the rules of constructing an energy cycle. While the second video shows how these rules are applied to construct the energy cycle.
The first video: A simplified guide to drawing an energy cycle.

The second video: Application of rules to an actual example.

-- -- -- -- --
Article written by Kwok YL 2009.
- If you would like to use this source, kindly drop me a note by leaving behind a comment with your name and institution. I am all for sharing as the materials on this blog is actually meant for the education purpose of my students.
- This material is entirely written by the author and my sincere thanks will be given to anyone who is kind, generous and gracious to point out any errors.
Friday, May 1, 2009
Ionic Equilibrium - Calculating constants.
(A) Acid Dissociation Constant.
A weak acid dissociates partially in water. Hence, the weak acid dissociates in a reversible equation thus an equilibrium is obtained. The experiment set up is depicted below.
 Using the equilibrium equation, we can create an ICE table (ICE = Intial, Change and Equilibrium) to determine the initial concentrations of the substances found in the equilibum, the change in concentration of the substances and the equilibrium concentration of these substances. We can do a direct substraction/addition between two concentrations because the volume of solution is the same (as shown in the above diagram).
Using the equilibrium equation, we can create an ICE table (ICE = Intial, Change and Equilibrium) to determine the initial concentrations of the substances found in the equilibum, the change in concentration of the substances and the equilibrium concentration of these substances. We can do a direct substraction/addition between two concentrations because the volume of solution is the same (as shown in the above diagram).
(B) Base Dissociation Constant.
The weak base dissociates partially, hence a weak base dissociates in a reversible equation thus an equilibrium is obtained. The set up is similar to that of a weak acid. Like in calculating the Acid Dissociation Constant, a similar ICE table is drawn. It is shown in the picture below. Do note that the concentration of water is not the variable as it is the medium where the solute dissolves in.

(C) Solubility Product.
This is applicable for ionic compounds that you learn are insoluble in water. Ionic compounds which are soluble in water will fully dissociate in water into its constituent ions. However, because of partial solubility due to interaction between solvent molecules and solid, this results in some amount of the ionic compound to dissolve in water. Hence, as a result despite a solid is insoluble in water, there are still traces of its constituent ions found in solution.
The diagram set up and the calculation is show below.
 As the ionic compound is a solid, concentration of a solid is not a variable.
As the ionic compound is a solid, concentration of a solid is not a variable.
-- -- -- -- --
Article written by Kwok YL 2009.
- If you would like to use this source, kindly drop me a note by leaving behind a comment with your name and institution. I am all for sharing as the materials on this blog is actually meant for the education purpose of my students.
- This material is entirely written by the author and my sincere thanks will be given to anyone who is kind, generous and gracious to point out any errors.
Homework 5 (due before 4th May 2009)
This week's question is as following observations:
(i) Explain why the bond energy of O-F is smaller than the bond energy of O-Br. (The former is 212 kJmol-1 and the latter is 217 kJmol-1.)
(ii)As working, draw the Lewis diagram of N2 and the CN- ion. You do not need to submit your Lewis diagram. (Clue: Draw HCN first)
Explain why the triple bond in N2 is stronger than the triple bond in CN-. (The former's bond energy is 944 kJmol-1 and the latter is 890 kJmol-1
Answers
The following pictures illustrate and explain the questions.
 Although, O-Br bond also have lone pair on both O and Br, the repulsion is much less because of Br's large size.
Although, O-Br bond also have lone pair on both O and Br, the repulsion is much less because of Br's large size. Comments
CommentsGenerally (i) has better answers. But, many missed out the key phrase - lone pair on O and the lone pair repulsion on F repel each other because they are so close together (due to O and F being small sized), hence weakening the O-F bond. - I think Samuel's answer for (i) is nice. Succinct, not trying to explain too much and doesn't complicate the situation further.
In addition, using electronegativity to explain (i) is NOT correct.
Interestingly, only Nicholas mentioned that overlapping between small atoms is weak because orbtial overlap is not effective. This is highly incorrect.
For (ii)
I think the biggest problem is that many of you tried to explain that lone pair repulsion, hence making the bond weaker. That is not true as illustrated by the picture above, where both CN- and N2 have lone pairs of electrons.
Hence, in short. The general strategy to explain strength of covalent bond is as follow.
(A) Direct application of the definition of covalent bonds.
If the two examples you are given cannot be concluded using (A), then either one of the two exceptions will be employed.
(B1) A polar covalent bond. A partial positive and partial negative charge, this is an added electrostatic attraction that strengthens the bond between the two atoms.
(B2) Lone pair repulsion. This causes the bond to be weaker.

