Predict with suitable explanation which compound has stronger inter-atomic bond, BeBr2 or AlBr3?
Answer
BeBr2 and AlBr3 covalent compounds. If both were ionic compound, both compound contains a very small and highly charge cation and a large anion. This results in the cation to polarise the anion's electron cloud, hence making electron clouds to overlap.
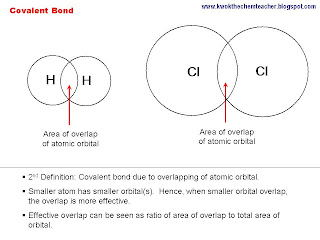
Since both BeBr2 and AlBr3 are covalent compounds, Be is a smaller atom than Al. Hence, the extent of overlapping of atomic orbital between Be and Br is greater than in Al and Br. Hence, Be-Br single bond is stronger than in Al-Br single bond.
Comments
The most comprehensive answer goes to Xing Ting, Johanan, Wei Jie and Dung. =)
The following are the misconceptions made. I have included the names so that we can all learn from each other. I think it wld be excellent if you would like to take this question into further discussion.
1. Stating that BeBr2 and AlBr3 are dative covalent compounds. Please don't say that. Covalent compound is good enough. - Eileen's answer
2. Not applying to the definition of covalent bond (key words missing out) completely. - Eileen's, Jo's, Daniel's
3. Thinking that BeBr2 are AlBr3 ionic compounds - Xiao Min's, Lyria
4. Incomplete sentence expression (with ambiguous word(s)). - Sherlyn (what is it?), Jing Yew (Am i going to assume that you meant covalent bonds are formed?)
5. Totally missing the point. Thinking that I am trying to account for dative bond or why covalent bond, when I am actually trying to determine strength of bond. - Peck Fen, Wen Han, Brandon
6. Writing irrelevant information (e.g. "Bond pair replusion", "more bonds bonds formed around Al, does not mean the bond is stronger", "VSEPR" and "Charges of Be and Al") - Renaldy, Benjamin, Crystal, Kevin, Nicholas
7. Trying to answer too much. Uses both definition of covalent bonding. (can get abit confusing). - Samuel